Custom phosphopeptides for kinase studies, phospho-specific antibody production, ELISA validation, dot blot assays, and signaling pathway research.
LifeTein provides custom synthesis of phosphorylated peptides, including single-site and multi-site phosphopeptides containing phosphorylated serine, threonine, or tyrosine residues. Phosphopeptides are widely used as kinase substrates, phospho-specific antibody antigens, assay controls, binding probes, and inhibitors in cell signaling research.
LifeTein offers phosphorylation on common protein signaling residues as well as selected D-amino acid phospho-residues.
Multi-site phosphorylation is also available, including peptides with two, three, four, five, or more phosphorylation sites depending on sequence length, solubility, and purification behavior.
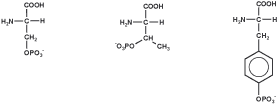
Phosphorylated peptides are technically more challenging than standard peptides because phosphate groups increase negative charge, alter solubility, and can reduce synthesis and purification efficiency. Multi-phosphorylated peptides require careful synthetic planning, optimized coupling conditions, and analytical confirmation.
LifeTein has synthesized a biotinylated 34-amino-acid peptide containing six phosphorylation sites:
[biotin]-xxxxxx[pSer][pSer][pSer]xxxxxxxxxxxxx[pSer]x[pThr][pSer]xxxxxxxx
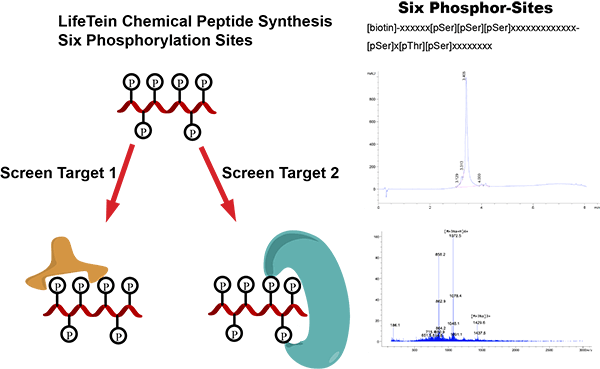
Phospho-specific antibodies are commonly generated using synthetic phosphopeptide antigens that contain the phosphorylated residue and surrounding native protein sequence. These antibodies are useful for detecting site-specific phosphorylation events in western blot, ELISA, immunohistochemistry, immunocytochemistry, flow cytometry, and other immunoassays.
For antibody production, LifeTein can synthesize:
Learn more about LifeTein polyclonal antibody services using phosphopeptide antigens →
Protein phosphorylation is one of the most important reversible post-translational modifications in cell signaling. Kinases add phosphate groups to serine, threonine, or tyrosine residues, while phosphatases remove them. This dynamic regulation can alter protein conformation, enzyme activity, localization, interaction partners, stability, and downstream signaling.
Because phosphate groups introduce strong negative charge, phosphorylation can act as a molecular switch. In cancer biology, immunology, neuroscience, metabolism, and cell cycle studies, site-specific phosphorylation often determines whether a signaling protein is active, inactive, localized, degraded, or bound to a specific partner.
Phosphorylation can be detected using several complementary methods. The best approach depends on whether the goal is to measure kinase activity, detect a known phosphorylation site, discover unknown phosphorylation sites, or quantify phosphorylation changes across samples.
Kinase activity can be measured by incubating a kinase with a defined substrate peptide in the presence of ATP. Phosphorylation is then detected using radioactive, colorimetric, fluorometric, luminescent, or mass spectrometry-based readouts.
Phospho-specific antibodies recognize a protein only when a particular residue is phosphorylated. Synthetic phosphopeptides containing the target phosphorylation site are commonly used as immunogens, while non-phosphorylated peptides are used for counter-screening and specificity validation.
Western blotting with phospho-specific antibodies enables detection of phosphorylated proteins in biological samples. This method is widely used for signaling pathway analysis and is often paired with total-protein antibodies for normalization.
Phospho-specific ELISAs provide more quantitative readouts than western blotting and are suitable for higher-throughput analysis. Cell-based ELISA can measure phosphorylation status in intact cells after stimulation, inhibition, or drug treatment.
Intracellular flow cytometry allows phosphorylation analysis at the single-cell level. Immunocytochemistry and immunohistochemistry can localize phosphorylated proteins within cells or tissues when validated phospho-specific antibodies are available.
Mass spectrometry can identify phosphorylation sites and distinguish phosphorylated peptide species. Because phosphopeptide signals may be less abundant than non-phosphorylated peptides, enrichment methods such as immobilized metal affinity chromatography, titanium dioxide enrichment, or phospho-specific immunoenrichment are often used.
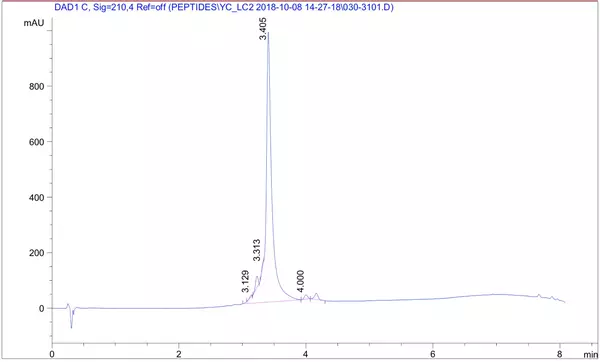
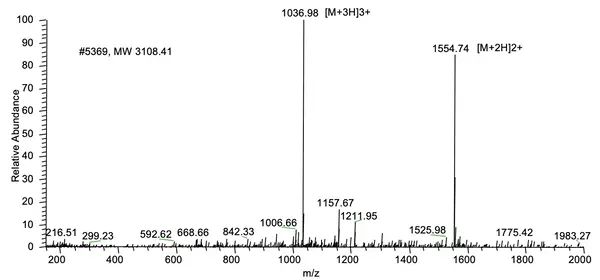
LifeTein synthesized a 26-amino-acid peptide containing five phosphorylation sites and delivered the peptide at 95.6% purity.
HPLC Results:
MS Results:
LifeTein designed and synthesized a series of phosphorylated peptides used for antibody development and validation. ELISA and dot blot analysis were performed to verify antibody specificity.
“Antibodies to the Thr345, Ser416, and Ser497 phosphorylation sites of XPO5 were generated in collaboration with Lifetein LLC...” Supplementary information
LifeTein helped design and synthesize phosphorylated peptides used for phospho-specific antibody production. The resulting antibodies were used to study EGFR-mediated phosphorylation of AGO2 and its role in miRNA maturation under hypoxia.
“The following peptides were chemically synthesized for antibody production in mice by Lifetein, ELISA verification, and peptide competition assay in immunohistochemistry...” Supplementary information
Please send your peptide sequence, phosphorylation site notation, desired purity, quantity, and any antibody-production or assay requirements. For phospho-specific antibody projects, we recommend including both the phosphorylated peptide and the matched non-phosphorylated control peptide.