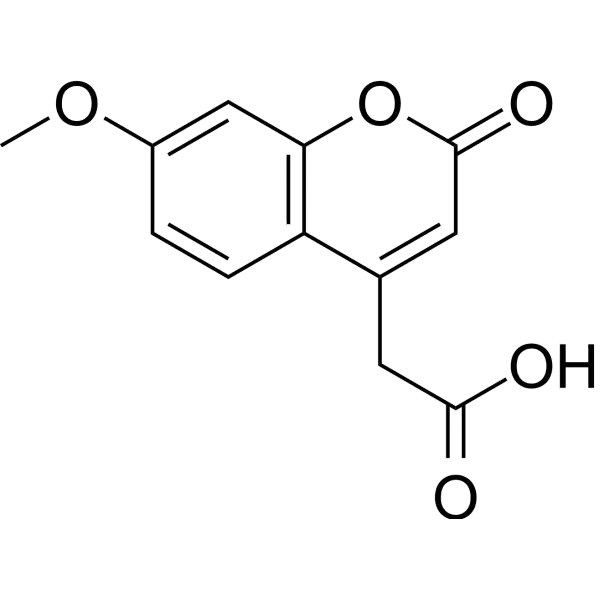
Fluorescent labelling is a cornerstone technique for visualizing and quantifying biomolecular interactions, with 7-Methoxycoumarin-4-acetic acid (MCA) standing out as a particularly versatile fluorophore for peptide applications. As a coumarin-derived dye, MCA is prized for its favorable photophysical properties and its specialized role in constructing sensitive, internally quenched substrates. Its primary utility lies in Fluorescence Resonance Energy Transfer (FRET)-based assays, where it acts as a donor fluorophore paired with a suitable quencher. This configuration allows for the real-time, continuous monitoring of enzymatic activity, making MCA-labeled peptides indispensable tools in protease research, drug discovery, and cellular biology. Companies like LifeTein provide expert synthesis of these complex probes, enabling researchers to tailor substrates for specific experimental needs.
Key Takeaways
- MCA is a coumarin-based fluorescent dye with excitation/emission maxima in the near-UV to blue spectrum, ideal for FRET applications.
- Its primary application is in creating internally quenched FRET substrates, where it is paired with quenchers like DNP (2,4-Dinitrophenyl) to measure protease activity.
- Conjugation to peptides is typically achieved via its succinimidyl ester derivative, allowing for stable attachment to the N-terminus or lysine side chains.
- These assays offer high sensitivity, real-time kinetic data, and the ability to work with nanomolar enzyme concentrations.
- Custom synthesis services, such as those from LifeTein, are crucial for producing sequence-specific MCA-peptide conjugates with high purity for reliable research outcomes.
Photophysical and Chemical Profile of MCA
Spectral Characteristics
MCA exhibits classic coumarin fluorescence, with absorption and emission peaks in the near-ultraviolet to blue light range. Reported maxima can vary slightly depending on the solvent environment; for instance, in methanol, peaks are observed at approximately 320 nm (absorption) and 380 nm (emission). In aqueous buffers and when conjugated to peptides, these values may shift, with common references citing excitation at 328-360 nm and emission at 393-410 nm. This spectral profile minimizes interference from biological autofluorescence, which is typically higher at longer wavelengths, thereby providing a low-background signal for detection.
Conjugation Chemistry
For practical use, MCA is activated as a succinimidyl ester (MCA-OSu), a reactive form that facilitates efficient conjugation to peptides. This chemistry targets primary amine groups, primarily the N-terminal α-amino group or the ε-amino group of lysine residues. The reaction forms a stable amide bond, incorporating the fluorophore directly into the peptide backbone. This site-specific labelling is critical for maintaining the peptide’s biological activity and for ensuring consistent fluorescence properties across batches.
Find out more about fluorescent peptides here.
Primary Applications in Peptide-Based Research
The Foundation of FRET-Based Protease Assays
The most significant application of MCA is in the development of FRET-based peptide substrates for protease analysis. In these constructs, MCA is covalently attached to one end of a peptide sequence that contains the specific cleavage site for a target enzyme. A quencher molecule, most commonly DNP, is attached to the opposite end. When the peptide is intact, the close proximity of the quencher absorbs the energy emitted by the excited MCA donor, resulting in fluorescence quenching. Upon protease cleavage, the physical separation of MCA and the quencher abolishes this energy transfer, leading to a dramatic increase in MCA fluorescence that is directly proportional to enzymatic activity.
A Practical Example: Monitoring Mitochondrial Protease OMA1
A clear illustration of this principle is found in research on the mitochondrial protease OMA1. To study this enzyme, researchers employed a custom peptide substrate: MCA-AFRATDHG-(Lys)DNP. This sequence contains the precise cleavage site of OMA1 within the OPA1 protein. In the intact peptide, DNP quenches MCA fluorescence. When OMA1 cleaves the peptide between arginine and alanine, the fluorescence is dequenched, providing a direct, spectrophotometric readout of OMA1 activity. This assay enabled the first direct activity measurements for OMA1, showcasing how MCA-labeled peptides can unlock the study of previously challenging enzymes.
Versatility Across Protease Families
The MCA/DNP pair is not limited to a single enzyme. It is a standardized tool for investigating a wide array of proteolytic enzymes, including matrix metalloproteinases (MMPs), caspases, and viral proteases. For example, a classic substrate for stromelysin (MMP-3) is MCA-Pro-Leu-Gly-Leu-DPA-Ala-Arg-NH₂. The modular design of these peptides allows researchers to easily swap the central cleavage sequence to target different proteases, making MCA a universal component in the protease researcher’s toolkit.
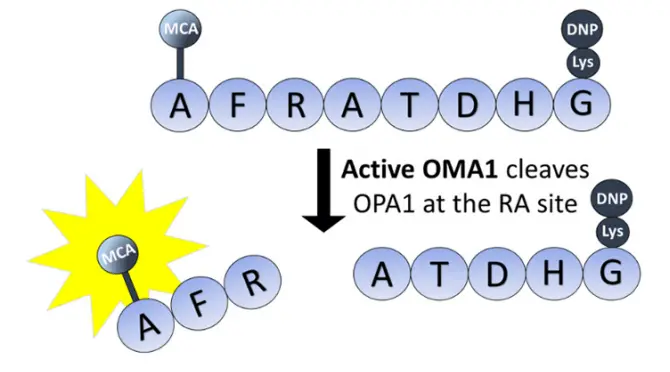
Considerations for Implementing MCA Labelling
Design and Synthesis
Successful assay development begins with careful peptide design. The cleavage sequence must be specific to the target protease, and the positioning of the MCA and quencher must ensure efficient FRET in the uncleaved state. Given the complexity of synthesizing and purifying these dual-modified peptides, partnering with a specialized provider like LifeTein is highly advantageous. Their expertise ensures high-purity products, which are essential for obtaining reliable, reproducible kinetic data and minimizing background signal.
Practical Assay Considerations
When running assays, researchers must optimize buffer conditions (pH, ionic strength) to support both enzyme activity and fluorescent signal stability. Establishing a standard curve with free MCA is necessary to quantify the amount of cleaved product. Furthermore, control experiments with enzyme inhibitors are crucial to confirming that the observed fluorescence increase is due to specific proteolytic cleavage.
Find out more about peptide synthesis here.
Frequently Asked Questions (FAQ)
What makes MCA particularly suitable for protease assays?
MCA is ideal because its emission spectrum overlaps strongly with the absorption spectrum of common quenchers like DNP, enabling highly efficient FRET quenching. Its photostability and the significant fluorescence increase upon cleavage make it excellent for sensitive, continuous kinetic measurements.
Can MCA be used for live-cell imaging?
While possible, MCA is less common for live-cell imaging compared to longer-wavelength dyes like GFP or Cy5. Its excitation in the UV/blue range can cause higher cellular autofluorescence and phototoxicity. However, it can be effective for in vitro or fixed-cell applications where its spectral properties are advantageous.
What is the difference between MCA and other coumarin dyes like AMC?
MCA (7-Methoxycoumarin-4-acetic acid) contains a carboxylic acid group for covalent conjugation to peptides. In contrast, AMC (7-Amino-4-methylcoumarin) is a cleavage product of many fluorogenic substrates but is not typically used for direct peptide labelling, as it lacks the same convenient reactive handle.
Are there alternatives to the MCA/DNP FRET pair?
Yes, several other FRET pairs are widely used. Common alternatives include EDANS/DABCYL (which operates at longer wavelengths) and FAM/Dabcyl. The choice of pair depends on the available instrument filters, the required sensitivity, and potential sample-induced interference.
Tobacyk, J., Parajuli, N., Shrum, S., Crow, J. P., & MacMillan-Crow, L. A. (2019). The first direct activity assay for the mitochondrial protease OMA1. Mitochondrion, 46, 1–5. https://doi.org/10.1016/j.mito.2019.03.001