LifeTein provides peptide conjugation services for projects that require selective, efficient linkage between peptides and other functional components such as PEGs, oligonucleotides, lipids, fluorophores, small-molecule payloads, nanoparticles, or proteins. Depending on the project, we use maleimide–thiol ligation, azide–alkyne click chemistry, copper-free SPAAC, tetrazine–TCO chemistry, and related site-selective strategies.
In practice, the most appropriate chemistry depends on the molecule pair, the desired site of attachment, the stability requirements, and whether the conjugation must occur under copper-free or highly orthogonal conditions. For many routine peptide conjugations, maleimide–thiol chemistry remains the simplest and most direct method when a defined cysteine is available. Click chemistry becomes especially useful when orthogonality, multifunctional assembly, or oligonucleotide compatibility is important.
| Maleimide–thiol | Often the most straightforward route for cysteine-containing peptides and PEG-, lipid-, or payload-bearing maleimide reagents |
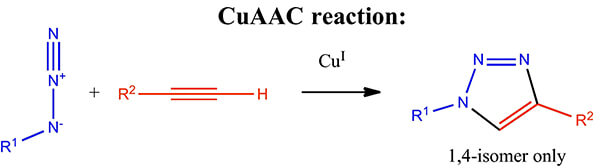
| CuAAC | Useful for azide–alkyne ligation in well-controlled synthetic settings |
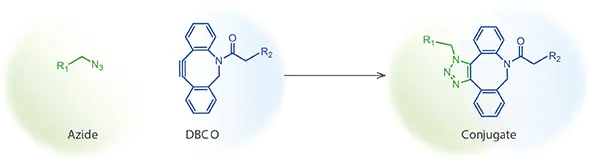
| SPAAC | Copper-free azide–cyclooctyne ligation for sensitive systems and bioorthogonal workflows |
| Tetrazine–TCO | Fast bioorthogonal ligation suitable for specialized dual-functional or in situ labeling concepts |
| Project types | PEGylation, oligonucleotide conjugation, peptide drug conjugates, peptide-lipid conjugates, and multifunctional research constructs |
Which chemistry is right for your project?
If you already know your preferred chemistry, we can work from that. If not, we can usually help decide whether a cysteine–maleimide route, PEG-linker route, SPAAC, CuAAC, or tetrazine–TCO strategy is the better fit for your construct.
Need a quote?