Fluorescent peptides play a crucial role in biological research, enabling the visualization and tracking of molecules and processes. Among the various fluorescent dyes available, Alexa Fluor 647 stands out due to its bright and photostable properties. Delve into fluorescent peptide labeling with Alexa Dye 647, exploring its applications, advantages, and considerations.
Introduction to Alexa Fluor 647
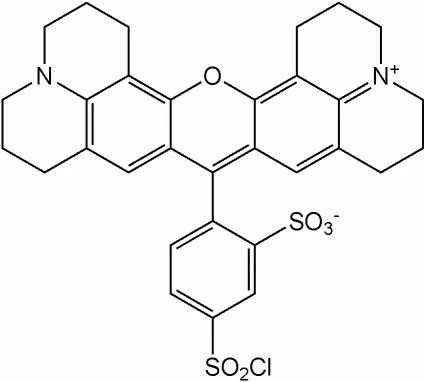
Alexa Fluor 647 is a succinimidyl ester dye that efficiently reacts with primary amines of proteins, forming stable dye–protein conjugates. Here are some key points about Alexa Fluor 647:
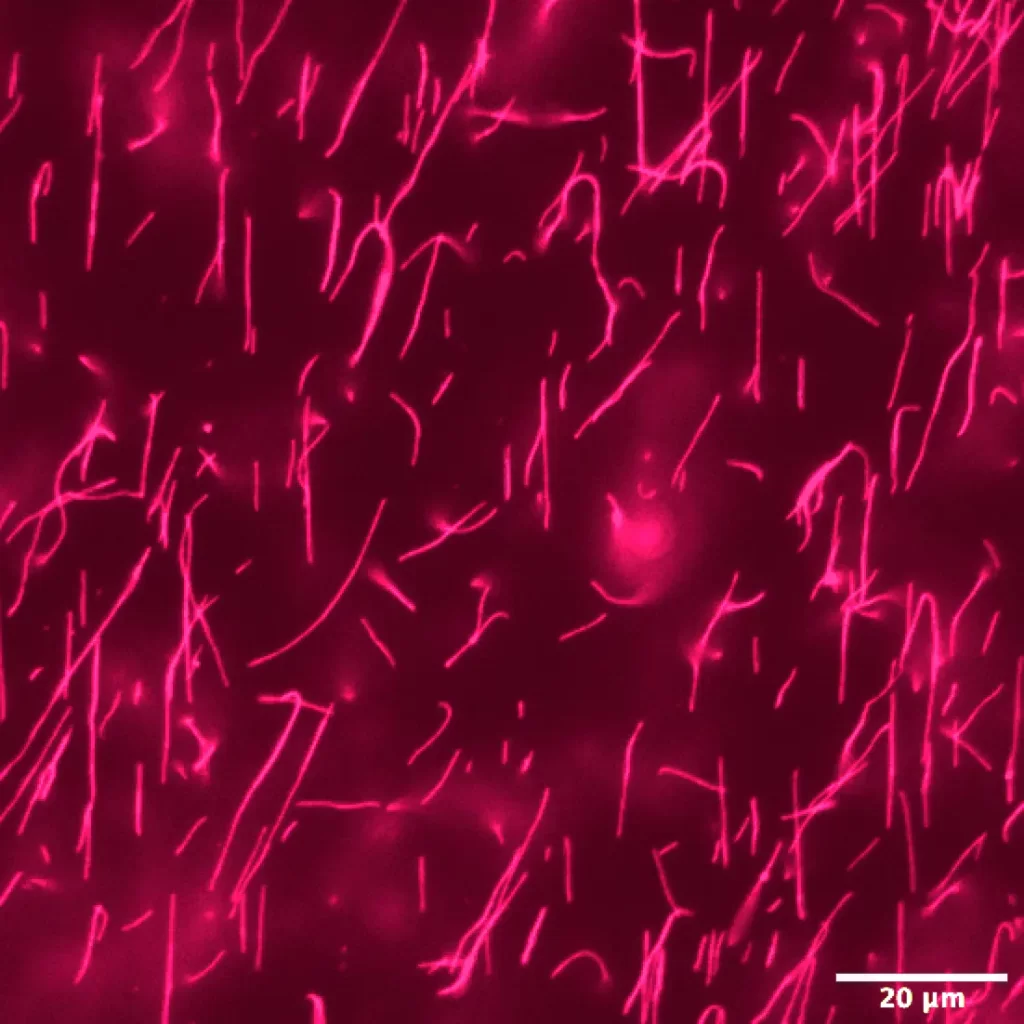
- Bright and Photostable: Alexa Fluor 647 emits light at approximately 668 nm, making it ideal for multicolor applications. Its wide spectral separation from other red fluorophores ensures minimal interference.
- pH Insensitive: The fluorescent signal remains consistent between pH 4 and 10, allowing reliable imaging across different cellular environments.
- Resistant to Quenching: Unlike Cy™5 dye, Alexa Fluor 647 maintains fluorescence even at high degrees of substitution.
Applications of Alexa Fluor 647-Labeled Peptides
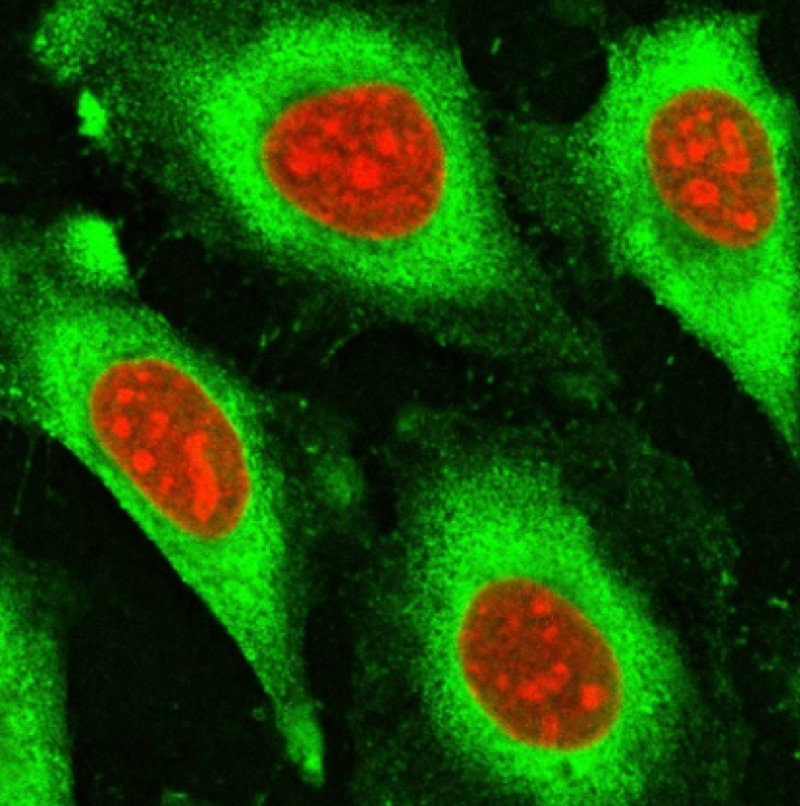
- Fluorescence Microscopy: Alexa Fluor 647-labeled peptides serve as excellent probes for visualizing cellular structures, protein localization, and dynamic processes.
- Protein-Protein Interactions: Researchers use these labeled peptides to study interactions between proteins, shedding light on signaling pathways and molecular networks.
- Enzymatic Activity Monitoring: By attaching Alexa Fluor 647 to specific peptide substrates, enzymatic activities can be tracked in real-time.
- FRET Assays: Fluorescence Resonance Energy Transfer (FRET) studies benefit from Alexa Fluor 647 as both donor and acceptor fluorophores.

Considerations and Best Practices
- Buffer Choice: To achieve optimal labeling efficiency, ensure the purified protein is in a buffer free of ammonium ions or primary amines.
- Protein Purity: Impure proteins, such as antibodies in crude serum, may not label well.
- Storage: Store Alexa Fluor 647 at ≤–20°C and protect it from light. Components B-D should be stored at 2–6°C.
- Fluorescent Dye Selection: Based on your specific experimental needs, consider other fluorescent dyes (e.g., FITC, Cy3, Cy5).
Practical Tips for Successful Peptide Labeling with Alexa Fluor 647
Conjugation Strategies
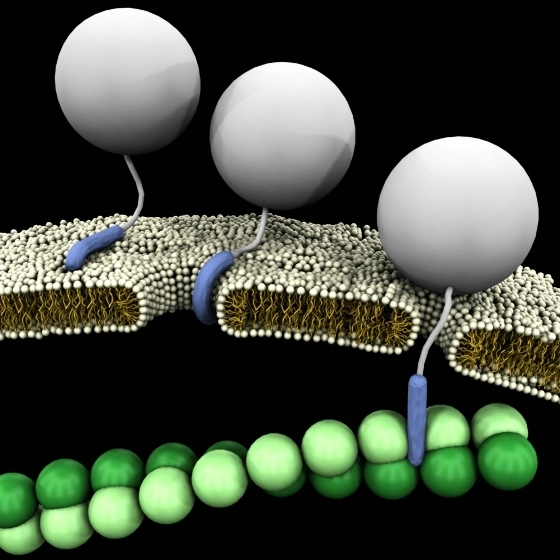
- Direct Labeling: In direct labeling, the dye is directly attached to the peptide. This method is straightforward but requires careful optimization of reactant ratios and reaction conditions.
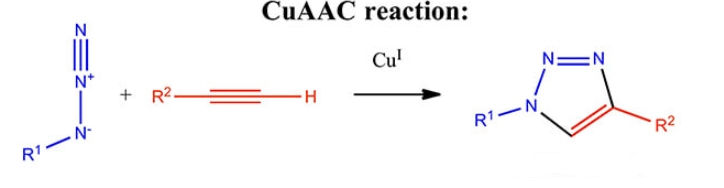
- Two-Step Labeling: Here, the peptide is first modified with a reactive group (e.g., Cys-Maleimide, Lys(N3), NHS ester) and then conjugated to Alexa Fluor 647. This approach allows better control over labeling efficiency.
Find more about Peptide Synthesis here.
Frequently Asked Questions
Q1: Can I use Alexa Fluor 647 for live-cell imaging?
A: Yes! Alexa Fluor 647 is suitable for live-cell imaging due to its photostability and minimal photobleaching. However, cell permeability and potential cytotoxicity should be considered.
Q2: What is the recommended storage condition for labeled peptides?
A: Store labeled peptides at –20°C in the dark. Components B-D should be stored at 2–6°C.
Q3: Can I multiplex Alexa Fluor 647 with other fluorophores?
A: Absolutely! Alexa Fluor 647 pairs well with other dyes like FITC, Cy3, and Cy5. Optimize spectral overlap for multicolor experiments.