The photocleavable biotinylated peptides can be selectively captured through biotin-streptavidin purification and cleaved or released under UV irradiation. For example, The photocleavable biotin azide peptide has a biotin linked to an azide group through a spacer arm containing a photocleavable moiety. The captured biomolecules can be efficiently photoreleased using a low intensity 365 nm UV lamp. Using Cu(I)-catalyzed Alkyne-Azide (CUAAC) or Cu(I)-free strain-promoted Alkyne-Azide Click Chemistry (SPAAC) reactions, the alkyne or DBCO conjugated molecules can be introduced to the azide containing photocleavable biotin peptides.
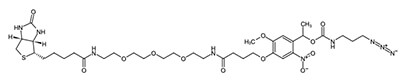
Biotinylated peptides are designed for screening ELISA (Enzyme-linked immunosorbent assay) assays that require immobilized peptides onto streptavidin coated 96-well plates, membranes, or glass slides.
The traditional methods are challenging to elute biotinylated peptides from immobilized avidin. It requires denaturation of streptavidin by boiling the resin or trypsin digestion of proteins. However, the photocleavable biotin peptides reduce the limitation of the streptavidin-biotin affinity purification. Captured biomolecules can be efficiently released at irradiation with low intensity UV lamp.
More Readings: Lifetein’s Photocleavable Linkers Assist Advancement of Microrobots in Anticancer Drug Delivery
References:
1. Proximity-dependent labeling identifies dendritic cells that drive the tumor-specific CD4+ T cell response, Science Immunology, 4 Oct 2024, Vol 9, Issue 100, DOI: 10.1126/sciimmunol.adq8843
2. Nakandakari-Higa S, Walker S, Canesso MCC, et al. Universal recording of immune cell interactions in vivo. Nature. 2024 Mar;627(8003):399-406. doi: 10.1038/s41586-024-07134-4.
3. Lee, C.S., Chen, S., Berry, C.T. et al. Fate induction in CD8 CAR T cells through asymmetric cell division. Nature (2024). https://doi.org/10.1038/s41586-024-07862-7
4. Pasqual, G., Chudnovskiy, A., Tas, J. et al. Monitoring T cell–dendritic cell interactions in vivo by intercellular enzymatic labelling. Nature 553, 496–500 (2018). https://doi.org/10.1038/nature25442
Applications for Peptide ELISA
Please click here for a biotinylated peptides synthesis service quote now!